[ad_1]
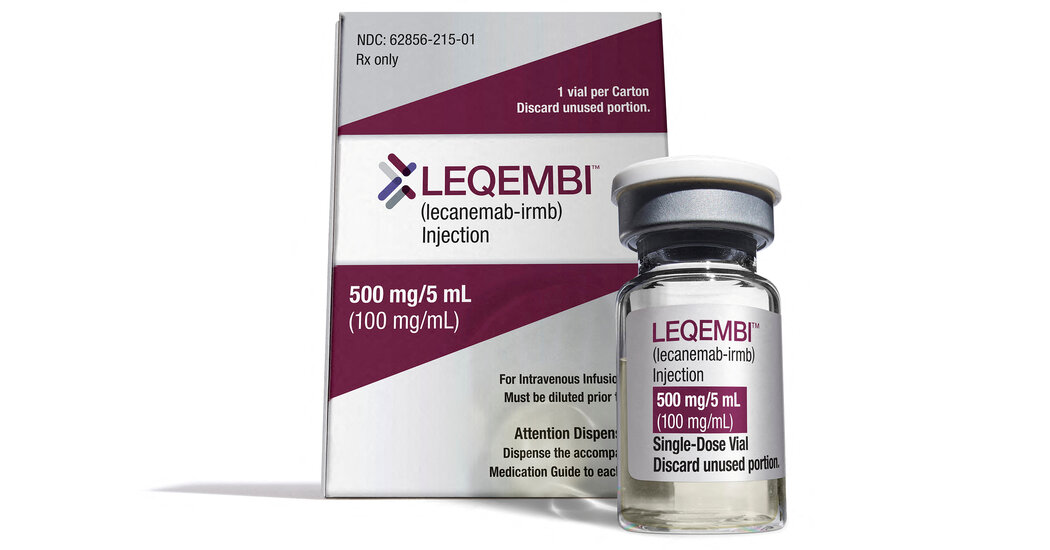
The Meals and Drug Administration on Thursday gave comprehensive acceptance to the Alzheimer’s drug Leqembi, and Medicare said it would include a great deal of its large price tag, laying the foundation for widespread use of a treatment that can modestly slow cognitive decrease in the early phases of the illness but also carries significant protection pitfalls.
The F.D.A.’s conclusion marks the very first time in two many years that a drug for Alzheimer’s has acquired whole approval, this means that the agency concluded there is stable evidence of likely profit. But the agency also additional a so-named black-box warning — the most urgent level — on the drug’s label, stating that in uncommon scenarios the drug can trigger “serious and everyday living-threatening events” and that there have been cases of mind bleeding, “some of which have been deadly.”
Leqembi can’t fix cognitive harm, reverse the study course of the disorder or halt it from obtaining even worse. But details from a massive medical demo implies that the drug — administered every single two weeks as an intravenous infusion — may well sluggish drop by about 5 months about about 18 months for people today with moderate signs or symptoms.
Still, some Alzheimer’s experts have said it is unclear from the health care evidence no matter if Leqembi’s skill to delay erosion of memory and cognition would be sufficient to be noticeable or meaningful for clients and their people. And whilst most instances of mind swelling and bleeding have been delicate or reasonable and have solved, there have been some major cases.
“The threats are really vivid,” mentioned Dr. Jason Karlawish, a co-director of the College of Pennsylvania’s Penn Memory Middle, who reported he will prescribe Leqembi after cautiously analyzing clients and explaining the probable pros and downsides. “Within the first couple months, you may well have little bleeds or swelling in your brain, which could or could not be symptomatic and if not detected in time can bring about disability.”
“In distinction,” Dr. Karlawish ongoing, “the advantages of slowing are delicate. You are not likely to working experience the notion of changes in your cognition or functionality in the exact same sum of time.”
Nevertheless Medicare will deal with 80 percent of Leqembi’s $26,500 price tag, patients could nonetheless shoulder hundreds of dollars in co-payments.
Eisai, a Japanese pharmaceutical company, led the advancement and tests of Leqembi (pronounced le-KEM-bee). Eisai is partnering and splitting profits with the American corporation Biogen, the maker of the controversial Alzheimer’s drug Aduhelm, for its commercialization and advertising.
The F.D.A.’s approval of Aduhelm was seriously criticized since the evidence of probable gain was inconclusive, with a single trial exhibiting modest slowing of drop but a different demonstrating no slowing. In advance of that approval, a committee of impartial advisers and an F.D.A. council of senior officials said there was not ample evidence that it labored. A lot of health-related facilities declined to prescribe Aduhelm, and Medicare has included it only for medical demo participants, sharply proscribing its availability.
Proof supporting Leqembi is much clearer, Alzheimer’s gurus said.
Leqembi will be readily available for persons with moderate dementia or a pre-Alzheimer’s affliction named mild cognitive impairment. The F.D.A. label instructs health professionals not to treat individuals with out screening to confirm they have an accumulation of the protein amyloid, a hallmark of Alzheimer’s that Leqembi attacks.
About 1.5 million individuals in the United States are believed to be in the commencing phases of Alzheimer’s. Numerous more — about 5 million — have progressed way too far to be eligible for Leqembi. Alex Scott, Eisai’s govt vice president of integrity, stated the firm endorses sufferers cease employing Leqembi as soon as they create moderate Alzheimer’s disorder.
Alzheimer’s industry experts mentioned they would advise some patients that they experienced greater chance for brain inflammation and bleeding — which includes those having blood thinners, these with additional than 4 microscopic bleeds in the mind and those with an Alzheimer’s-connected gene mutation identified as APOE4.
The threat to men and women with two copies of the APOE4 mutation — about 15 p.c of people today with Alzheimer’s — is so large that the F.D.A.’s black-box warning endorses that all people be genetically examined to assess their basic safety possibility and spells out that those people with two APOE4 copies are far more vulnerable to establishing “symptomatic, critical and severe” mind bleeding or swelling.
The black-box warning will apply to all drugs that, like Leqembi, are monoclonal antibodies that assault amyloid. Leqembi is the initial to get total approval, but other folks are in many levels of progress.
The warning does not point out clients who are using blood thinners, but Leqembi’s label claims that “additional caution really should be exercised” when looking at whether or not to give blood thinners to Leqembi people.
The F.D.A. greenlighted Aduhelm less than a system referred to as “accelerated approval,” which can be provided to drugs with unsure gain under unique criteria, which include that the company conduct a further clinical demo. Leqembi been given accelerated acceptance in January, but that position intended Medicare would only include the drug in confined conditions.
The F.D.A. selection granting comprehensive acceptance to Leqembi suggests that Medicare will address it for suitable patients.
Nevertheless, some clients will be not able to manage the 20 p.c Medicare does not cover, perhaps about $6,600 a calendar year. Such as expenditures of professional medical visits and required typical brain scans, some of which will obtain Medicare reimbursement, the cure could run to about $90,000 a yr, some authorities estimate.
A current review estimated that covering the drug and essential services for about 85,000 individuals would cost Medicare $2 billion a 12 months and would climb to $5.1 billion if the selection of clients achieved about 216,000. That could lead to an raise in premiums for all Medicare beneficiaries, not just those people receiving Leqembi, the study explained.
In interviews, Ivan Cheung, the chairman and main government of Eisai’s United States functions, approximated that in the first three several years, about 100,000 patients would be getting the drug.
The Medicare company is including a need that medical professionals prescribing Leqembi post health care details about just about every individual right before and though they are being addressed with the drug. The info will be retained in affected person registries and evaluated to discover much more about Leqembi’s advantages or harms, the agency claimed.
“With F.D.A.’s selection, C.M.S. will cover this treatment broadly whilst continuing to assemble data that will assistance us recognize how the drug works,” the administrator of the Centers for Medicare and Medicaid Products and services, Chiquita Brooks-LaSure, reported in a assertion.
Some advocacy groups, like the Alzheimer’s Association, have criticized the registry necessity, calling it an pointless barrier to access. But healthcare gurus say registry programs are widespread and effortless to comply with. Their concern is that the registry will not be evaluating Leqembi clients with many others, so it will not be capable to say if Leqembi slows cognitive drop.
The F.D.A.’s approval on Thursday was dependent on a significant demo indicating that patients acquiring Leqembi declined 27 p.c far more slowly and gradually around 18 months than sufferers getting a placebo. The variation involving people acquiring drug and placebo was compact — a lot less than 50 % a position, on an 18-place cognitive scale that assesses features like memory and challenge-fixing. Some Alzheimer’s professionals say that for slowing of drop to be clinically meaningful, or obvious to individuals and households, the difference in between the groups should be at minimum one point.
Leqembi sufferers also declined extra slowly but surely on 3 secondary steps of cognition and daily purpose, and info on biological markers was typically more powerful for Leqembi than for the placebo. All these actions shifting in the very same route strengthens the strategy that the drug can profit individuals, gurus say.
Even now, a report on the data, revealed in The New England Journal of Medicine and co-composed by researchers from Eisai, concluded that “longer trials are warranted to figure out the efficacy and safety.”
Worries about protection have been stoked by reports of deaths of 3 scientific demo contributors who professional mind inflammation and brain bleeding, two of whom had been staying taken care of with blood thinners. Eisai has said it is unclear if Leqembi contributed to their fatalities because the patients experienced complex clinical troubles.
“You’ve bought little rewards and a specified hazard for major adverse events, and that has to be well balanced,” reported Dr. Lon Schneider, director of the California Alzheimer’s Illness Heart at the University of Southern California, who explained he will prescribe Leqembi to carefully evaluated sufferers.
“If its efficacy ended up higher, we would not be conversing about adverse activities as a lot for the reason that we would see a apparent gain,” he claimed, adding, “I assume several people today will see this and say it is not value the effort and hard work, it is not worth twice-a-month infusions.”
Dr. Karlawish explained the conclusions dealing with clients and people will be challenging. Because qualified sufferers have only moderate symptoms of cognitive decrease, some may decide to just take any treatment that may extend that comparatively useful phase, though other individuals might only think about the challenges of the drug worthwhile if they were a lot additional impaired.
Dr. Karlawish claimed a single latest affected individual declined to get evaluated for opportunity treatment method, indicating that “‘I want much more benefits, I really do not see the benefit.’” But, he reported, “I have other patients, nevertheless, who would say, You necessarily mean you can give me a drug that could gradual the disorder?”
In the trial, approximately 13 % of patients getting Leqembi skilled brain swelling, which was mostly gentle or average, while considerably less than 2 percent of individuals getting the placebo knowledgeable these kinds of swelling. Most brain inflammation did not cause any symptoms, generally emerged before long immediately after use started and solved within just a couple of months. About 17 percent of Leqembi people professional brain bleeding, as opposed with 9 p.c of clients receiving a placebo. The most popular symptom from brain bleeds was dizziness.
General, the benefits suggest the danger of mind bleeding and inflammation was appreciably decrease than for individuals in trials of Aduhelm.
Dr. Jerry Avorn, a professor of medication at Harvard Healthcare School who experiments treatment regulation and use, claimed medical practitioners will sense pressure to prescribe Leqembi from patients, people and advocacy companies. Medical establishments will also have an “enormous economical incentive” for the reason that of the Medicare reimbursement that “they could then spend on social staff and all the other points that Medicare will not reimburse,” he reported, incorporating “any economically self-respecting memory middle is heading to see this as an financial windfall.”
[ad_2]
Source hyperlink