[ad_1]

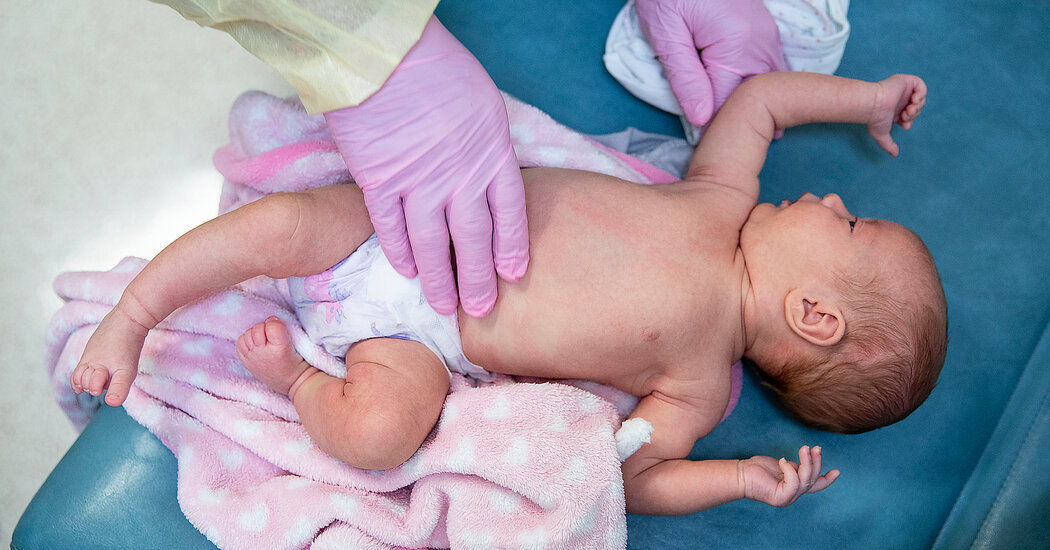
RSV is a seasonal virus that lands tens of countless numbers of youthful youngsters in the hospital just about every calendar year. On Thursday, advisors to the Fda voted in favor of approving a extensive-performing antibody that protects infants from RSV.
Christoph Soeder/Photo Alliance by means of Getty Photos
disguise caption
toggle caption
Christoph Soeder/Image Alliance via Getty Images

RSV is a seasonal virus that lands tens of countless numbers of youthful kids in the clinic each individual 12 months. On Thursday, advisors to the Food and drug administration voted in favor of approving a long-acting antibody that protects infants from RSV.
Christoph Soeder/Image Alliance by way of Getty Images
A panel of advisers to the Food stuff and Drug Administration have advised that the agency approve a new antibody drug to protect infants from major lung health problems brought about by respiratory syncytial virus, also recognised as RSV.
On Thursday, the panel voted in favor of Fda approval for the injectable antibody medicine – identified as nirsevimab – immediately after several hours of testimony from the drugmaker AstraZeneca, Food and drug administration scientists and the community. The dilemma before the panel was regardless of whether the added benefits of the cure outweigh the risks.
The drug, if authorized, would offer infants protection from the virus in their initial RSV seasons with a single shot. It would be much more cost-effective and a lot more greatly offered than the solitary current preventive drug – a monoclonal antibody shot referred to as palivizumab – which calls for regular monthly administration and is reserved for infants at high medical hazard.
There was unanimous assistance on the 21-man or woman committee for approving the drug’s use in infants ahead of or during their very first RSV period. And, in a different vote, all but two associates of the panel supported providing the drug to infants with professional medical hazards by their 2nd RSV period.
“This is just one of the most vital infectious conditions in the pediatric population,” reported Dr. Mary Anne Jackson, a pediatrician at Children’s Mercy in Kansas Town, Missouri and a voting member.
Customers of the committee claimed the facts offered by the drugmakers, AstraZeneca and Sanofi, and the Fda shown that nirsevimab appears to be protected and powerful.
RSV is a viral infection that places between 58,000 to 80,000 younger little ones in the clinic each yr, creating it the primary result in of hospitalization among infants in the U.S. According to AstraZeneca’s analysis, most RSV hospitalizations could be prevented with use of this drug.
“This is a pathogen that has a sizeable influence on the lives of young small children, resulting in significant morbidity and mortality” and limits the potential of children who are sick with other illnesses to get health-related treatment, says Dr. Steven Krug, a pediatric crisis medical professional and professor at the Northwestern University Feinberg School of Medication.
In two scientific trials, involving nearly 3,000 toddlers, a solitary shot of the monoclonal antibody decreased the dangers of an RSV scenario that expected health care interest by 70-75%, and lowered the hazards of hospitalization by 60-80%. Substantial levels of the antibody persisted for at least five months. Aspect effects, including rashes and fevers, ended up unusual and generally gentle.
“Total, all committee users had been amazed with the carry out of the examine and the clear results that ended up presented,” stated Dr. Lindsey Baden, an infectious health conditions health practitioner at Brigham and Women’s Clinic in Boston and chair of the FDA’s Antimicrobial Medications Advisory Committee, “The committee believes this is an crucial advance.”
Although the Fda isn’t required to adhere to suggestions, it normally does. The agency is anticipated to come to a decision in the 3rd quarter of 2023.
If permitted, the CDC’s Advisory Committee on Immunization Techniques is envisioned to weigh in with suggestions on how the drug should be employed in distinct populations. The businesses say they are prepared to start the preventive shot in the U.S. ahead of the 2023-2024 RSV period, if the item clears the regulatory course of action by then.
It’s expected to be priced comparable to a “premium vaccine system” and would be offered beneath the commercial title “Beyfortus,” said Jon Heinrichs, an executive at Sanofi. The antibody was authorised last slide by authorities in the EU, and is currently remaining viewed as for use in quite a few Asian nations.
The Food and drug administration is also thinking of other medication and vaccines in growth that would enable secure toddlers from RSV. They are expected to situation a selection on an RSV vaccine for expecting mothers by late August.
[ad_2]
Resource url