[ad_1]
Why It Matters: R.S.V. can be deadly
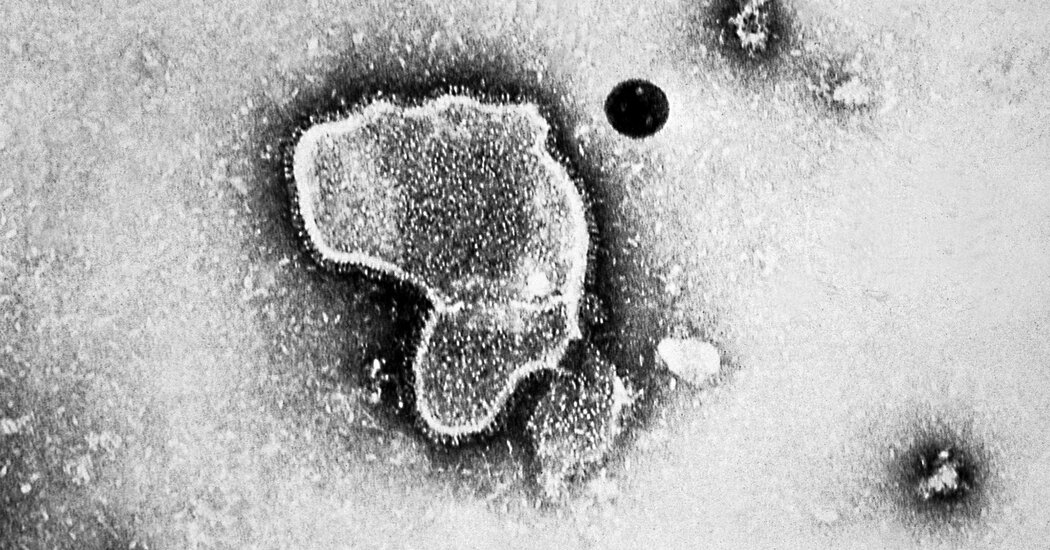
The F.D.A. estimates that R.S.V. is related with 6,000 to 10,000 deaths every calendar year in adults 65 and older and at least 60,000 hospitalizations in that age team. It is a leading killer of small children around the world.
This winter season, R.S.V. contributed to the “tripledemic” also involving flu and Covid conditions that swamped children’s hospitals and some I.C.U. wards.
Saying the approval, Dr. Peter Marks, the agency’s vaccine division chief, explained: “Older older people, in particular people with fundamental health ailments, these kinds of as heart or lung condition or weakened immune techniques, are at substantial possibility for extreme condition triggered by R.S.V.”
Qualifications: Added benefits and risks of the shots
On March 1, an F.D.A. advisory panel reviewed facts from trials for two R.S.V. vaccines aimed at older adults, a single from GSK and a person from Pfizer. The panel suggested that the company approve each.
The GSK vaccine was virtually 83 % productive in stopping lessen respiratory tract disease in adults 60 and older in a examine of about 25,000 clients, in accordance to details published in The New England Journal of Medication. The virus can guide to pneumonia, which is significantly a lot more worrisome for the elderly.
Pfizer’s R.S.V. vaccine for more mature older people is also predicted to acquire F.D.A. approval this thirty day period. In a significant analyze of that shot, it was observed to be almost 67 per cent efficient in preventing R.S.V.-related disease.
The Pfizer and GSK vaccines had been even far more helpful in treating older and sicker sufferers.
The advisers did understand of some exceptional facet outcomes from the vaccine trials. In the days soon after the pictures had been specified, two recipients of the Pfizer vaccine and one particular recipient of the GSK shot created circumstances of Guillain-Barré, a affliction where the immune procedure attacks the nervous procedure, in accordance to facts introduced to the F.D.A. panel.
The moment the pictures grow to be accessible to the community, the agency stated it would require GSK to keep an eye on the incidence of Guillain-Barré and yet another unusual ailment that was perhaps related to the shot.
Moderna is also establishing an R.S.V. vaccine for this age team, and reported it expected authorization in the very first half of this calendar year. A trial of 37,000 more mature adults showed 82 p.c efficacy of its shot, the business explained, with “no safety issues identified” while analyses ended up continuing.
AstraZeneca and Sanofi are trying to get F.D.A. approval of a monoclonal antibody treatment method to shield infants and toddlers up to 2 a long time aged from R.S.V. bacterial infections. Conclusions of a big review showed the treatment diminished confirmed ailments by 75 % right after 1 shot, in accordance to AstraZeneca.
Pfizer has used for independent acceptance of an R.S.V. vaccine to be supplied in the later on levels of pregnancy to safeguard youthful infants.
What is Next: When will the shots be out there?
It will still be months in advance of the adult vaccine is publicly readily available in the United States.
The Centers for Ailment Manage and Avoidance is predicted to observe the F.D.A.’s approval, most possible issuing its suggestion in June.
GSK said its vaccine would then be accessible in the tumble at U.S. pharmacies, clinics and other well being care configurations.
GSK executives have claimed that provides of the vaccine, which is produced generally at a plant in Belgium, should really be easily readily available. For Medicare people with Aspect D drug protection, there would be no out-of-pocket price, Alison Hunt, a GSK spokeswoman, stated. But the firm has not produced a price tag, even though insurers generally address substantially of the value of lots of vaccines.
Last 7 days, the European Medicines Company advised acceptance of GSK’s vaccine for grown ups 60 and older. The corporation claimed it hoped the photographs would also be approved later for use in Japan and China.
[ad_2]
Supply link